We often hear the claim “More regulation is better.” But is it? Like everything else in society, regulations have a cost. Excessive regulations that delay the approval of an innovative product can cost firms tens of millions in lost revenue and deprive consumers of a lower-cost option. Regulatory uncertainty can also lead to firms shifting their investments to other, more “regulatory-efficient” countries.
The rejection of agricultural innovations in Europe, especially genetically modified (GM) crops, has cost its economy billions in lost investment opportunities. In January 2012, the German-based agricultural and chemical giant BASF announced that its Europe-based Plant Science Division would move to the United States because of delays in regulatory decisions in Europe. In part, this decision was influenced by the 13 years it took to receive European approval for a GM potato variety developed by BASF. This relocation has cost the European Union (EU) several hundred jobs and tens of millions annually just in BASF’s research and development (R&D) investments. The other multinational seed development companies have all followed similar courses of action.
Billions in agricultural innovation investment has fled Europe, largely because of its regulatory system’s utter failure to render consistent and timely approval decisions for GM crops. Currently, less than one-tenth of global agricultural R&D budgets is invested in the EU; 20 years ago, the share was one-third, according to Bayer CropScience’s Julian Little. The EU regulatory system for GM crops has approved only two varieties for commercial production since 2003. Even decisions on approval for animal feed imports take longer in the EU than in the US: an average of 995 days in the EU, compared with 686 days in the US. The EU is saying to the global agriculture R&D industry, “Don’t invest here,” a message that has been heard loud and clear.
The time required for regulatory approval of GM crops has consistently increased in other regions as well. The first GM traits to be regulated were for herbicide tolerance and insect resistance in 1994. Over the next decade, the length of the US Department of Agriculture regulatory process more than doubled for these same two traits: between 1994 and 1999 the approval process took 5.9 months, and between 2000 and 2004 it took 13.6 months. A 2011 report found the average approval time for a GM crop variety in the US had risen to 25 months. Globally, the average number of months required for a GM crop variety to receive regulatory approval in 2011 was 65 months, up from 49 months in 2008.
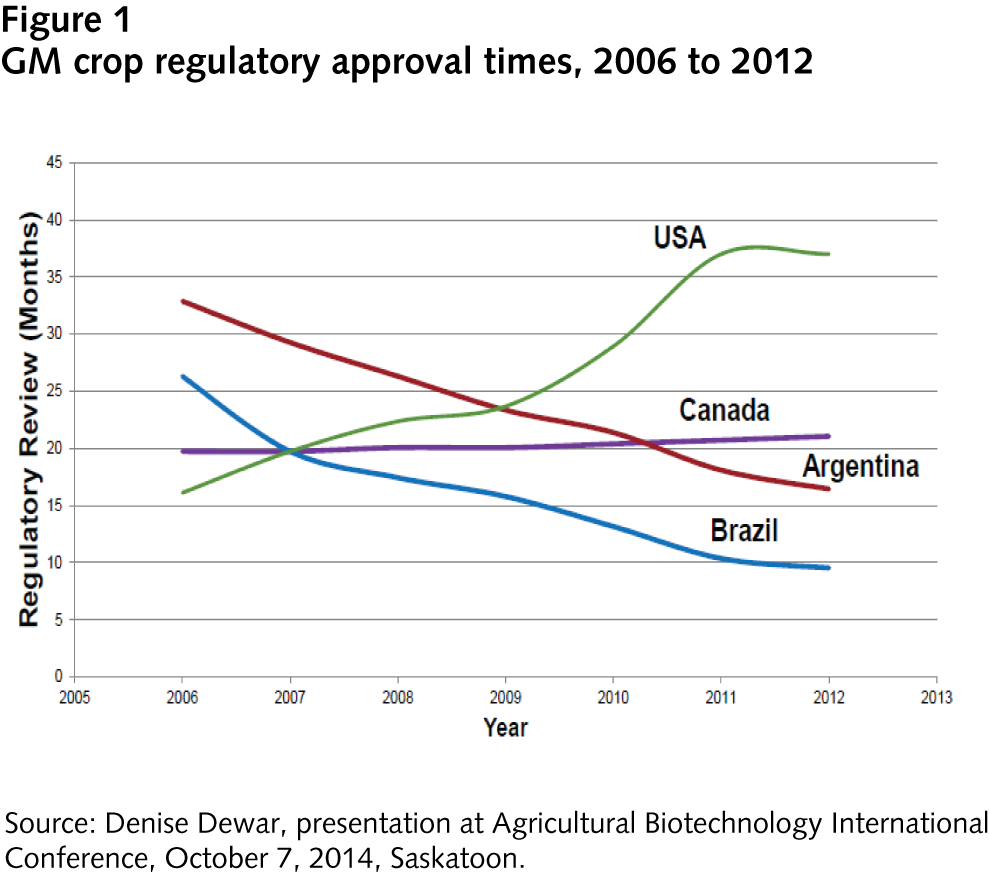
Figure 1 illustrates the regulatory approval speed for the four leading GM-crop-producing nations from 2006 to 2012. Canada’s regulatory approval times have marginally trended upward since 2006, to just over 20 months.
This trend could be viewed with concern in light of the regulatory approval process for two non-browning GM varieties of Golden Delicious and Granny Smith apples. In 2011, Okanagan Specialty Fruits submitted these apple varieties to the Canadian regulatory agencies; the apples were not granted approval until March 2015. Canadian regulators took nearly twice as much time as had been needed for other GM product approvals. However, concerns about increasing regulatory approval times were downplayed following the approval of Simplot’s new GM potato varieties with decreased acrylamide (a carcinogenic compound that develops in potatoes when they are baked or fried at high temperatures). Health Canada and the Canadian Food Inspection Agency (CFIA) received the dossier from Simplot early in 2015 and provided approval for four varieties of Innate potatoes in May 2016. This approval within 15 months suggests that Canadian regulatory agencies may be increasing their efficiencies.
No one would advocate that diligence in risk assessment be sacrificed for private sector investment. But after safely regulating GM crop varieties for over 20 years, Canada’s regulatory system should focus on the efficiencies that have been gained. Canada has established itself as a global leader in the risk assessment of GM crop varieties, having reviewed and approved them since 1995. Over this time, Canadian scientific research and regulatory review have yet to identify a single environmental or human health concern.
Perhaps now is the time to review whether herbicide-tolerant and insect-resistant GM varieties of canola, corn and soybeans — unlike new varieties developed through conventional plant breeding — require continuing regulatory oversight after initial approvals. These GM varieties are no longer “new”: they are now the conventional varieties, since they are grown by over 90 percent of Canadian farmers, according to our research. Removing the additional oversight for more GM varieties of these three crops would allow Health Canada and the CFIA to redirect resources toward assessment of new varieties of other crops.
Without substantial increases in funding, it will be challenging for Health Canada and the CFIA to keep approval times consistent with the promising pattern of recent years.
While GM crops face increased scrutiny in some countries and additional regulations add greater costs, the innovative science of biotechnology continues to expand. New gene editing techniques are rapidly being applied to many varieties of agricultural products in public and private R&D laboratories in Canada and elsewhere, which means the quantity of varieties being submitted for approval is about to increase dramatically. Is it realistic to expect that our regulatory agencies will be able to process a wave of new submissions and still meet their current obligations? Without substantial increases in funding, it will be challenging for Health Canada and the CFIA to keep approval times consistent with the promising pattern of recent years.
As gene editing technologies become more widely adopted and adapted, they have the potential to revolutionize plant breeding. Governments that choose to rigorously regulate these technologies, following the EU model of GM crop regulation, will undoubtedly lose innovation investments. The built-in uncertainty of this type of regulation is enough to influence firms as they decide where to invest. But countries that regulate the products of gene editing technologies as scientific evolutions of current technologies will likely benefit from these investments, and additionally from the innovative products once they are commercialized.
In a world of ever-advancing science, the ability of a country’s regulatory system to deliver consistent and timely decisions has become the determining factor for investments in innovation. Canada would gain an agricultural R&D advantage by treating plant varieties developed via gene editing technologies the same way as it treats the products of conventional plant breeding, without an extra layer of regulatory oversight. Canada should learn from the EU’s experience and adjust its approach so that we get the most out of the scientific revolution to come.
This article is part of the Canadian Agriculture at the Cutting Edge special feature.
Photo: Shutterstock/Alex Traksel
Do you have something to say about the article you just read? Be part of the Policy Options discussion, and send in your own submission. Here is a link on how to do it. | Souhaitez-vous réagir à cet article ? Joignez-vous aux débats d’Options politiques et soumettez-nous votre texte en suivant ces directives.