Putting Canada’s intellectual property (IP) architecture on a sounder footing is critical to the potential of the Canadian pharmaceutical industry.
The inadequacy of the current IP regime in the pharmaceutical sector also constitutes a point of contention in the current negotiations towards the Comprehensive Economic and Trade Agreement (CETA) with the 27 nations of the European Union (EU). Canada is being urged by EU negotiators to strengthen its intellectual property framework. Three specific changes they are calling for are a robust appeals process for innovative pharmaceutical companies, patent term restoration, and extended protection of data from innovative pharmaceutical companies’ clinical drug trials.
CETA negotiations offer Canada an important impetus to modernize its pharmaceutical IP regime. CETA could significantly increase bilateral trade with Europe and provide a boost of billions of dollars to the Canadian economy. However, modernizing Canada’s IP protection for pharmaceutical products could mean more than reducing trade frictions and expanded access to foreign markets. The proposed reforms offer the opportunity to increase investment and employment in this thriving sector and to further encourage much needed innovation.
Although Canada currently enjoys great benefits from the pharmaceutical industry, including jobs and investment, the industry is characterized by a research and development process that is lengthy, expensive, uncertain and risky. Regardless of varying degrees of overestimation or underestimation of the cost of pharmaceutical research and development, it is clearly a tremendously costly endeavour. In their 2007 article “The Cost of Biopharmaceutical R&D: Is Biotech Different?,” J. A. DiMasi and H. G. Grabowski estimated the cost of developing a new medicine at US$1.3 billion and, according to the Pharmaceutical Research and Manufacturers of America (PhRMA), new drug development takes an average of 10 to 15 years with no guarantee of success.
The potential for free riding on the fixed costs borne by the innovating firm constitutes a market failure that would discourage most innovation in the absence of intellectual property protection. Patents and the other forms of intellectual property rights protection address this market failure, providing innovators with a limited period of market exclusivity. Society balances market exclusivity to encourage innovation against public access to this knowledge. Through this trade-off, the existing patent system corrects the market failure that would stymie most innovation. Patent protection is disproportionately more important for the pharmaceutical industry than virtually any other to ensure that the innovator realizes the returns on research and development (R&D).
Overall, empirical evidence suggests that stronger intellectual property protection for pharmaceuticals stimulates additional investment in R&D and innovation. The preponderance of studies examine patent protection, thus less is understood about the importance of other types of protection and the elements that should characterize other legislation. While IP protection is clearly an essential national strategy, it is crucial to recognize that the pharmaceutical industry is global in nature and laws in one country may affect the health of the industry in another. In a 2011 article Patricia Danzon pointed out that through parallel trade the weak patent protection in one nation “effectively spills over to other countries, undermining the ability of the manufacturer to realize the value of the patent in countries that respect patents.”
The potential for free riding on the fixed costs borne by the innovating firm constitutes a market failure that would discourage most innovation in the absence of intellectual property protection.
Canada’s complacency in IP protection is threatening its relative position in the global pharmaceutical industry and changes are needed to re-establish Canada as a global competitor. Adjustments need to be made to patent term extensions, price regulation of patented drugs and basic patent linkage with automatic injunction. Updating our IP laws would provide the necessary incentives for innovation and open up trade relationships worth billions. Canada should implement patent term restoration, which would provide innovative pharmaceutical firms up to five years of additional product exclusivity to compensate for the lengthy regulatory approval process, as well as extended protection for the data from the clinical trials of innovative pharmaceutical companies.
By adopting global legislative best practices, specifically those that characterize innovation-intensive nations, Canada could attract additional pharmaceutical research and development investment and gain the research jobs that would follow. However, any changes in legislation should preserve the existing system’s commitment to safety and efficacy, ensuring the health of patients above all.
In Canada, the legal framework surrounding pharmaceutical intellectual property relies on three components. The primary intellectual property law enacted by the federal government is the Patent Act, which sets the patent regime in Canada. The Patented Medicines (Notice of Compliance) Regulations (NOC Regulations) provide for patent linkage, balancing the interests of innovator companies with those of generic manufacturers. Finally, the Food and Drug Regulations protect the data exclusivity of innovators.
Patents for pharmaceutical products and processes provide for 20 years of exclusivity for an invention disclosed in the patent application. The criteria for the invention are such that it must be novel, useful and non-obvious. Given the complexity of pharmaceutical innovation it is common for a single drug to embody many technologies and be protected by many patents with distinct expiry dates.
The EU’s concerns about a right of appeal for innovative pharmaceutical companies against generic producers are related to the issue of “patent evergreening.” Critics point to “patent evergreening” as a strategy to obtain multiple patents on the same product, essentially extending the term of exclusivity presented in the original patent grant. While it may be argued that incremental innovations will contribute less to social welfare than innovations that are both first-in-class and best-in-class, follow-on innovations are nonetheless important advances and worth encouraging. Optimal patent regimes will reward subsequent innovations and also allow original innovators to capture a share of the returns from incremental innovations that were spurred by the initial technological advance. A recent study by the Congressional Research Service notes that since much technological innovation occurs incrementally, incremental innovations may provide significant benefit to patients and promote competition.
The NOC Regulations connect the regulatory approval of generic drugs and patents. Prior to bringing a generic drug to market, the generic manufacturer must address the patents asserted to be relevant by the innovator company before Health Canada will issue marketing authorization. The generic firm may await expiry or claim the relevant patent is invalid or not infringed. The result is that generic firms may first face a summary proceeding to determine patent validity and later risk litigation if infringement is claimed. In like manner, the innovating firm may face litigation under the NOC Regulations and also in defending a patent’s validity. Through the patent linkage, the NOC Regulations seek to balance the incentives for innovation and the timely arrival of generic competition to the market.
Innovative drugs are protected from generic competition by Canadian law for a period of eight years through the protection of innovator data under the Food and Drug Regulations. As Paul Grootendorst and Aiden Hollis point out in their paper “The Canada-European Union Comprehensive Economic and Trade Agreement: An Economic Impact Assessment of Proposed Pharmaceutical Intellectual Property Provisions,” the “Minister of Health cannot grant a market authorization to a product that would directly or indirectly rely on the clinical trials sponsored by the firm that obtained the regulatory approval.” This exclusivity does not apply to new indications for existing drugs and only applies to drugs meeting certain criteria, specifically the first chemical entity launched in Canada.
In cross-country comparisons of patent regimes, it is most useful to examine both the most innovative nations and the emerging markets that show the greatest promise for future innovation.
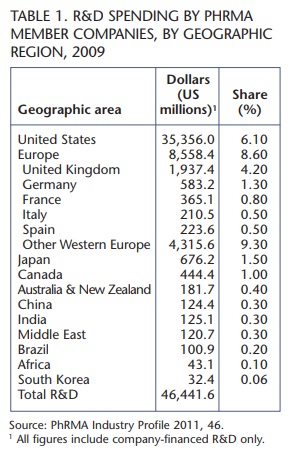
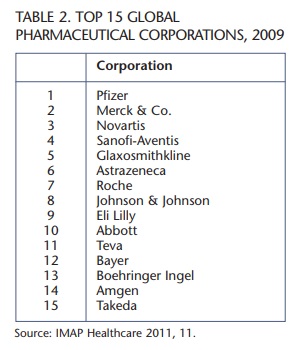
In order to identify the nations garnering the greatest share of research and development dollars, table 1 presents the figures for research and development spending by geographic region for PhRMA member companies in 2009. It is worth noting that of the top 15 global pharmaceutical corporations in 2009 shown in table 2, only Roche and Teva are not PhRMA member companies.
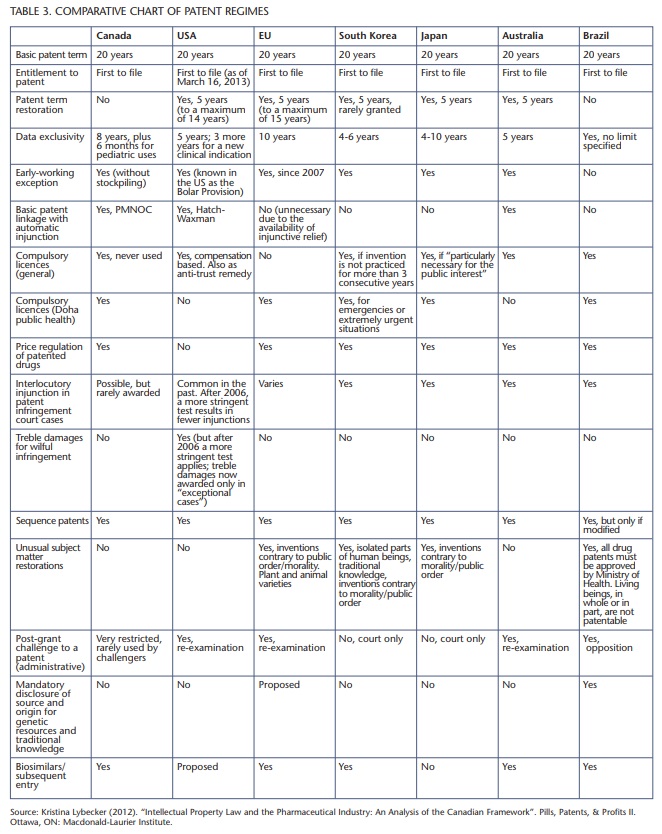
The most significant differences across regimes appear to be in the areas of patent term extensions (where Canada is an outlier without legislation), price regulation of patented drugs (where the United States is an outlier without legislation) and basic patent linkage with automatic injunction (where Canada, the United States and Australia have provisions in place while the European Union, South Korea and Japan do not). Table 3 provides perspective on how different regimes compare and contrast with one another and how Canada measures up across each type of legislation. The countries with the highest levels of research and development spending done by PhRMA member companies, as described in table 1, include the United States, several Western European nations and Japan. Accordingly, these are the legal environments most worth exploring.
Four changes in Canadian legislation are recommended based on legislative differences across the seven countries with the highest levels of research and development spending by the world’s largest innovative pharmaceutical companies: Canada, the United States, the European Union, South Korea, Japan, Australia and Brazil.
In stark contrast to the United States and the European Union, Canada fails to provide an extra period of patent protection as compensation for time lost during regulatory approval delays. While Canadian law provides for a 20-year patent term, standard under the Agreement on Trade Related Aspects of Intellectual Property Rights (TRIPS), there is no provision for the reduction in effective patent life due to the lapse between the filing of a patent and the grant of market authorization.
The restoration of a patent term extension of zero to five years, as in other countries, would lengthen the effective patent life of innovative therapies, increasing the incentives to invest in the research and development costs that these treatments require.
Canada’s complacency in IP protection is threatening its relative position in the global pharmaceutical industry and changes are needed to re-establish Canada as a global competitor.
Currently, while innovative drugs are protected from generic competition through the protection of innovator data for eight years, only drugs meeting certain criteria are eligible. In addition, data exclusivity currently does not apply to new uses for existing drugs in Canada. While data exclusivity regimes differ across countries in nature, scope and extent of protection, jurisdictions such as the EU and United States provide innovative pharmaceutical companies with broader protection than Canada. The EU provides for data exclusivity extensions of 8+2+1 years. Although the United States provides five years of data exclusivity with eligibility for an additional three years for data exclusivity limited to new and essential clinical trials, it also provides 12 years of data exclusivity for new biologics. Canada should provide for more expansive data exclusivity protection, including new uses, not just “innovative drugs.” Beyond such protection for small-molecule drugs, additional protection should also be provided for biologics. Strengthening Canadian data exclusivity laws will provide incentives for innovative firms to produce the data required for regulatory approval, facilitate the research process and encourage additional investment.
Canada should strengthen its anticounterfeiting legislation. Criminal sanctions in concert with regulatory provisions will protect Canada from the threat of fraudulent medicines, safeguard the health of patients and defend the innovative pharmaceutical industry from the theft of intellectual property.
Pharmaceutical counterfeiting both reduces the incentives for innovation and endangers public health and safety. Fraudulent medicines divert potential sales from innovator companies to criminal enterprises and expose the originator companies to legal liability.
Recently the Council of Europe adopted the Medicrime Convention on pharmaceutical counterfeiting and similar crimes involving threats to public health. As noted by the European Federation of Pharmaceutical Industries and Associations “White Paper on the Anti-Counterfeiting of Medicines,” the convention aims to strengthen product protection measures, ensure reliability in the wholesale distribution of pharmaceuticals and define clear obligations for starting materials. The World Health Organization’s IMPACT Programme echoes these recommendations. Suggestions included in the IMPACT Handbook (2011) include combating Internet distribution of counterfeit pharmaceuticals, establishing guidelines for a rapid response plan, developing good security practices for packaging materials, developing pharmacovigilance systems and identifying regulatory and legislative gaps.
An effective intellectual property protection framework will include legislation mandating significant criminal sanctions for pharmaceutical counterfeiting. As outlined by the European Federation of Pharmaceutical Industries and Associations (2010), legislation should prevent pharmaceutical products with false identities, histories or sources from entering the legal supply chain. Such protection of the industry’s research and development investments would promote public safety and encourage additional investments and further innovation.
Canada is one of the few industrialized nations lacking a policy for rare diseases, and the country should implement orphan drug legislation. Such legislation would allow Canadian firms to quickly get new therapies for the treatment of rare diseases to patients.
Unfortunately, the existing patent system fails to provide incentives for research into orphan (rare) diseases and diseases of the poor due to a lack of profits. As there is no official Canadian definition of rare diseases, it is worth looking to other nations for statistical perspective on these conditions. In the United States, rare diseases are those affecting fewer than 200,000 Americans, while the European Union defines such diseases as those affecting 1 per 2,000 or fewer people. Overcoming this obstacle to drug development for these types of diseases would enhance global social welfare and provide the potential for therapeutic relief to many of the world’s most vulnerable.
Pharmaceutical firms rely disproportionately on patents and other forms of intellectual property protection to ensure innovators are able to see returns to their research and development. Pharmaceutical patents protect process and product innovations, encouraging the development of therapies that enhance and extend lives on a global scale. This innovation necessitates protection, and this protection necessitates a trade-off. Patents provide market exclusivity in exchange for continued investment in innovation.
Canada’s current IP protection regime is a stumbling block to accessing billions of dollars on EU markets. Compared to Canada, EU countries are able to approve new drugs more quickly and provide earlier access to innovative medical therapies. With these recommended changes, Canada could attract more life science research investment, create additional research jobs and speed the development of new medicines.
The legislation that facilitates the growth of a prospering generic industry differs from that which nurtures a robust innovation-based industry. The recommendations described here provide for an enhanced legal environment to safeguard patient health, encourage therapeutic advances for rare diseases and foster a robust innovative pharmaceutical industry. While these suggestions may generate fierce debate, the discussion surrounding these proposed changes is critical to improving health in Canada and to sustaining the excellence of the Canadian health care system.
Photo: Shuttestock
This summary article is drawn substantially from “Intellectual Property Law and the Pharmaceutical Industry: An Analysis of the Canadian Framework,” Lybecker’s contribution to The Economics of Intellectual Property Protection in the Pharmaceutical Sector, the second instalment in MLI’s Pharmaceutical Series.